Unlocking the potential of 3d printing for spare parts availability

Creating a new supply channel can be problematic. Creating a new channel with an experimental technology, in the context of a waning global pandemic and persistent supply chain disruptions, is also challenging. But the potential benefits demand the attention.
The Background
During the height of the pandemic, a large pharmaceutical manufacturer was exploring ways to reduce supply chain costs without impacting plant reliability. The company had already learned that Original Equipment Manufacturers (OEMs) do not maintain inventory of specific spare parts, but instead supplies those parts on an as-needed basis. That meant long lead times under the best of circumstances. Manufacturers were carrying these long lead time spare parts in inventory to avoid downtime while absorbing all associated costs.
Additive manufacturing, or 3D printing, was suggested as a viable option to eliminate those long lead times and reduce those costs by its third-party MRO supply chain partner, RS Integrated Supply.
While 3D printing has been available for more than 20 years, it exploded in popularity since the pandemic. Some of that explosive growth is materializing along the MRO supply chain where companies are looking to build resilience around spare parts and tools needed to maintain production.
The Challenge
The risk-adverse client recognized the existing OEM lead times were unsustainable, with at least one supplier having an average lead time of six months. As the pandemic deepened, that lead time, and others, ballooned, opening consideration to a substitution process.
Through additive manufacturing, the company was looking to build an alternative manufacturing solution by mitigating supply shortages and risks.
Project Objectives
- Improve availability of identified critical spares with long lead times
- Reduce inventory
- Reduce purchasing costs and Total Cost of Ownership
- Maintain and improve plant reliability
- Create a 3D digital library of qualified items for additive manufacturing
The Solution
Like many industries, pharmaceutical manufacturers have strict change management protocols. For this client, for any substituted part to gain approval it must meet defined specifications and determined to be “Fit for Use.” To achieve this approval, the process requires a parts evaluation based on operational environment – temperature, humidity, vibration, stress, etc. – and criticality. Each of those tolerances must be built into the 3D printing plan, to ensure the proper material gets used and the correct print orientation is set. Other factors considered include, the type of material to be used for the product, slice height, or deposit thickness for each layer of material, fill density, and support materials used to hold the developing part in place.
Clear, quantitative guidelines from which to evaluate these conditions and a part’s Fitness for Use are required to avoid inaction simply from fear to change.
OEM relationships add an additional layer of complexity to the 3D printing process. There needs to be clarity between the client and its OEM suppliers regarding intellectual property around spare parts, warranties, and services. Questions to resolve before 3D production can begin, included: Are the parts protected by patents? Are the parts only available from the OEM supplier? Are they considered a commodity? Can they be fabricated? It is also important to document the impact on equipment warranties and services.
Some OEMs are embracing 3D printing as an alternative source for specific spares. Appropriate file types are often sold to the end user for printing, some with licences limiting the number of printed parts. Others will share the CAD files, handing off printing responsibility to the end user.
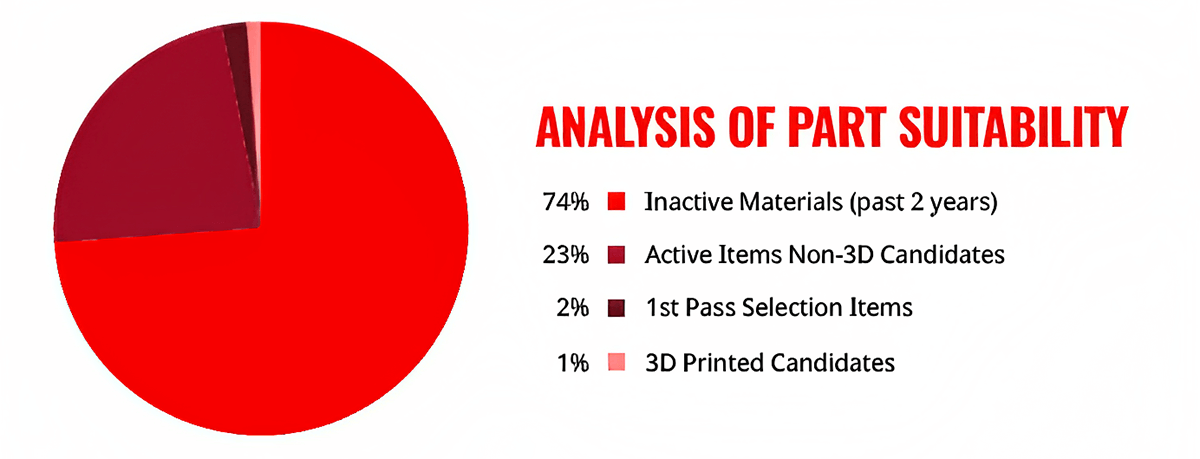
With all planning steps aligned and client agreement, RS Integrated Supply launched the project with an audit of the company’s inventory. The results showed more than 12,000 items in inventory. More than 9,000 of those spare parts were inactive, with no purchase activity in the previous 24 months. Of the remaining 3,000+ parts, more than 150 were determined eligible for 3D printing once any required digitalization was complete.
Digitalization involves creating an executable file of the part. It involves scanning the part to generate a point cloud image. That is followed by modelling the part to create the proper print format file, which directs the printing operation. During the modelling phase, scan imperfections are removed, and any requested design changes are made.
RS Integrated Supply has the experience and institutional knowledge to lead a global project to enrich MRO data and parts in order to develop a sustainable database. Their project management is excellent, the team is responsive and flexible, and they showed their capability in working through issues unique to specific sites across the globe. They interact directly with the subject matter experts at all global sites providing education, guidance, and encouragement to meet timelines.
The Result
An initial proof of concept project scoped 12 parts from inventory to review. The client was taking a cautious approach before committing to a full 3D printing program. Each of those 12 products shared similar characteristics:
- Single source, stocked item
- Special configuration required
- Mechanical replacement part
- OEM part
- Non-product contacting
The selected parts delivered a 17% to 85% reduction in piece price, excluding any costs for one-time digitalization and prototyping.
Lead time, meanwhile, saw an even larger return with 67% to 95% reduction in lead time, after technical data specifications received approval.
The results were more than expected, clearly proving the viability of the concept, with the client agreeing to expand the project. Phase Two involved 157 3D printed parts, with substantial lead time improvement and cost reductions.
Again, the company saw significant savings. Phase two saw more than $95,000 in material savings with an additional $64,000 reduction in inventory costs. Another $16,000 was earned in holding cost avoidance.
In total, nearly $200,000 in savings from just 157 parts.

Supply and Assets. In SyncTM.
Find out more about the savings we deliver for clients.

